Product Paramete
Catalogue NO. | MHW-WFK-C202309004 |
CAS No. | 9067-32-7 |
Product Name | Hyaluronic Acid |
Chemical Structure | 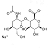
|
Application | Pharmaceutical excipients grade,Eye drop grade,Injection grade |
Package | 1kg net per foil bag,10kgs net per carton |
Appearance | White powder |
Molecular weight | 100MDa-250MDa |
Solubility | Water soluble |
Function | Moisturizing agents |
Shelf life | 2 years |
Storage | Keep container tightly closed and in a cool place. Keep away from heat. |
Dosage | 0.05-0.5% |
Application
Hyaluronic Acid the sodium salt of hyaluronic acid, is a linear high molecular weight mucopolysaccharide composed by thousands of repeating disaccharide units of D-glucuronic acid and N-acetyl-D-glucosamine.
1) High safety
Non-animal origin bacterial fermentation
A series of safety tests carried out by authorized testing or organizations
2) High purity
Very low impurities (such as protein, nucleic acid and heavy metal)
No pollution of other unknown impurities and pathogenic microorganism in production process assured by strict production management and advanced equipments.
3) Professional service
Pharmaceutical excipients grade -It has high polymer inertness, good biocompatibility and degradability
The medical applications include: eye moisturizer, wound auxiliary treatment, ophthalmic surgery auxiliary drugs, sprains, burn auxiliary treatment, etc.
Eye drop grade -It can lubricate, moisturize, improve the efficacy, relieve dry eye, and promote the healing of corneal and conjunctival injury
In the medical application: eye drops, eye ointment, ophthalmic surgery auxiliary drugs, etc
Injection grade -It has the effects of joint surgery filling, drug sustained release, lubricity, viscoelasticity, promoting cartilage repair, inhibiting inflammation, and relieving pain
The medical applications include: eye surgery, intra-articular injection surgery, intradermal injection, cartilage (joint) and tendon disease treatment


